
Instead, they readily share electrons via covalent bonds.Ĭommonly, carbon atoms share with other carbon atoms, often forming a long carbon chain referred to as a carbon skeleton. 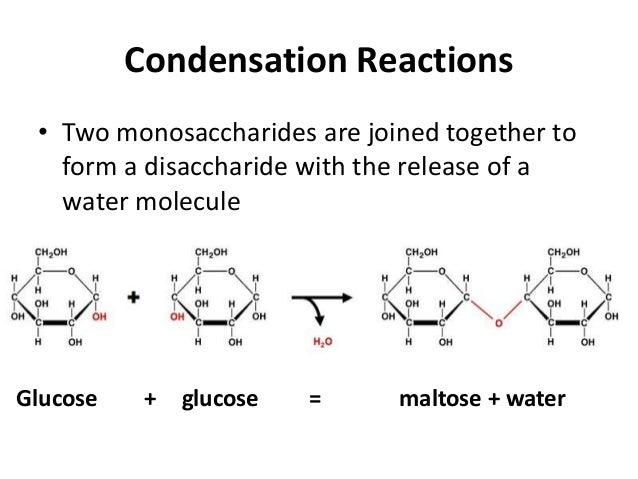
Carbon atoms do not complete their valence shells by donating or accepting four electrons. Recall that carbon atoms have four electrons in their valence shell, and that the octet rule dictates that atoms tend to react in such a way as to complete their valence shell with eight electrons. What makes organic compounds ubiquitous is the chemistry of their carbon core. Before exploring these compounds, you need to first understand the chemistry of carbon. The four types most important to human structure and function are carbohydrates, lipids, proteins, and nucleic acids. 
They are found throughout the world, in soils and seas, commercial products, and every cell of the human body. Organic compounds typically consist of groups of carbon atoms covalently bonded to hydrogen, usually oxygen, and often other elements as well. Identify the building blocks of nucleic acids, and the roles of DNA, RNA, and ATP in human functioning.Describe the structure of proteins, and discuss their importance to human functioning.Discuss four types of lipids important in human functioning.Provide examples of three types of carbohydrates, and identify the primary functions of carbohydrates in the body.Explain the chemistry behind carbon’s affinity for covalently bonding in organic compounds.Identify four types of organic molecules essential to human functioning.By the end of this section, you will be able to:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
